Regenerative Medicine & Stem Cells
Regenerative Medicine & Stem Cells
Regenerative medicine converges biomedical research, innovative technology and patient care – with the goal of using our body’s own resources to regenerate and repair damaged tissues.

What is Regenerative medicine? uitklapper, klik om te openen
Regenerative medicine is an area of biomedical research, technology and patient care that is rapidly growing. It aims to develop novel therapies to help our bodies restore or regenerate organs and tissues that have been damaged by injury, ageing, cancer and disease. We currently rely on organ transplants and artificial devices, but these do not restore original tissues. Our bodies contain stem cells, which are capable of regenerating damaged tissues and we hope that a wide variety of diseases can be treated either through cellular therapy or by stimulating the body’s own stem cells into repairing their own tissues.
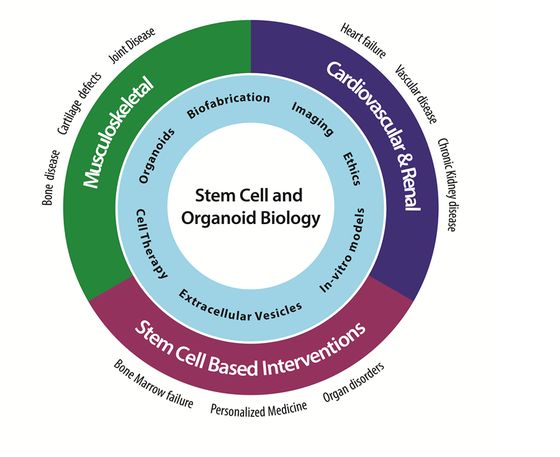
Research themes uitklapper, klik om te openen
Medical care uitklapper, klik om te openen
Regenerative medicine aims to develop new treatments that take advantage of the body's ability to repair damaged tissue itself. We strive to improve the quality of life with the help of modern treatments for people with illness, body wear, injury or bad genes. We do this by combining science and technology, bringing researchers and doctors together and striving to help our bodies recover themselves.
Education and training uitklapper, klik om te openen
Education and training young talent is a focus of Regenerative Medicine & Stem Cells. Through a variety of programs we aim to attract, train and educate the next generation of investigators and caregivers to develop and implement regenerative therapies.
Regenerative Medicine Center Utrecht uitklapper, klik om te openen
In a unique initiative, the UMC Utrecht and the Hubrecht Institute facilitated the creation of a newly built location that brings together the largest number of researchers in the field of regenerative medicine and stem cells in the Netherlands. This one of the few centers of such magnitude in Europe.
Chair regenerative medicine uitklapper, klik om te openen
Prof. dr. Marianne Verhaar is Professor of Experimental Nephrology and Head of the Dept. Nephrology & Hypertension. She is chair of the program Regenerative Medicine and Stem Cells.

Contact uitklapper, klik om te openen
Uppsalalaan 8
3584 CT Utrecht
The Netherlands


Social relevance uitklapper, klik om te openen
Researchers must often consider how to integrate innovation with social responsibility. Prof.dr. Annelien Bredenoord, Professor in Medical Ethics and member of the Regenerative Medicine & Stem Cells strategic program, investigates ethical aspects of research and research processes: “We work with researchers to identify and address ethical issues, and we develop guidelines or policy. In this way we contribute directly to the development of socially responsible and relevant treatments.”