Biological discovery heralds new medication

Proteins that regulate nerve cells bind to that cell's receptor to give their instructions. It was thought that when two of those proteins arrive at the same time, either one protein or the other binds. But this is not the case, according to research by UMC Utrecht and Oxford University: instead of competing, the signaling proteins both bind. This temporarily "switches off" the cell, which offers opportunities for all kinds of new therapies.
He thinks it is a great find, one that actually requires a reprint of the cell biology textbooks. Jeroen Pasterkamp, Professor of Translational Neursciences, has been researching proteins that control nerve fibers and nerve cells for some time. Jeroen: “These signaling proteins have all manner of functions. They act, for example, as a kind of traffic controller for the nerve cells, which make their way through the body during their development. They do this by binding to the receptor of a nerve cell - the landing platform as it were - and sending a signal that directs the cell in a certain direction. This creates healthy nerve pathways, but also determines, for example, how a tumor grows and spreads.”
Cell biology
Because there are quite a few of these types of signaling proteins - roughly a hundred - and because they continuously bind to nerve cells, Jeroen and his research team wondered what happens when multiple proteins arrive at the receptor at the same time. “We already knew that the proteins we are currently working with bind in the same place. The guiding principle in cell biology is that there is competition and there is a winner: either one or the other protein binds. We were particularly interested in that competition. We thought that if we had a better understanding of how that process works, we might be able to manipulate it to address a number of disease states at the molecular level.”
Large protein complex
To that end, the research team at the Oxford University laboratory first made large amounts of the receptor and ligands (the signaling proteins) and then put them together to make crystal structures. Jeroen: “To our surprise we saw that all those proteins bind to each other. Instead of competing with each other, they create a large protein complex comprising 2 ligands and 1 receptor.” They were even more astounded when they saw the biochemical result: no signal was passed on; in fact, nothing really happened anymore. “That means that the receptor effectively switches off,” says Jeroen, “and the cell becomes insensitive to the signals.” They published on this newly discovered biological principle together with colleagues from Oxford University in the journal Cell, in an article entitled: ‘Simultaneous binding of Guidance Cues NET1 and RGM blocks extracellular NEO1 signalling’.
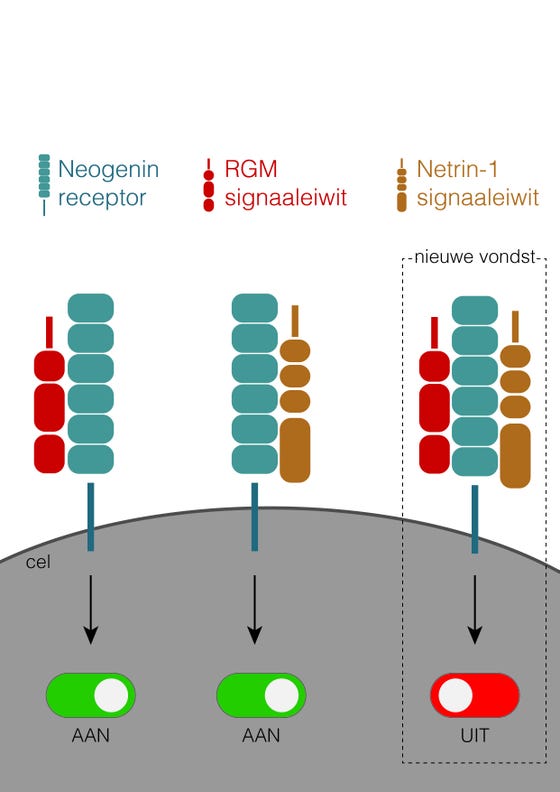
The research shows that the signaling proteins RGM and Netrin-1 do not only bind separately to the (Neogenin) receptor of a cell, but can also bind both at the same time, with the result that the receptor "switches off" and no signal is transmitted anymore.
General biological principle
While they only investigated a single combination of receptor and ligands for the scientific publication, Jeroen believes it is a general biological principle. “We are not entirely sure yet - for that, we need to make crystal structures of other complexes first - but we are also working with other receptor-ligand complexes in the lab and they all look very similar. At first, we did not understand how two ligands can bind to the receptor at the same time, but this soon became clear when we gained more insight into the 3D structure of the receptor-ligand complex.”
What is more, the fact that the receptor "switches off" has a logical function. Nerve fibers, for example, are designed to make contact with other nerve cells. That means that they first have to grow towards the cell and then stop moving. Jeroen: “It was not yet known that this 'stopping' happens this way, according to this new principle. But because nobody expected this outcome, we also started functional testing in the nervous system, with all kinds of manipulated proteins that we can make. Then we saw that it is true that if two ligands arrive at the same time, no signals are passed on and nothing happens at all."
Brain damage
The research initially focused on the effect of the principle in a healthy situation, but it is now clear that the outcome is potentially very interesting for the development of new medicines against various diseases. “We have collected so much information about protein interactions that we can start manipulating them,” says Jeroen. “Take an adult's nerve system, for example. There is hardly any recovery in case of damage to the brain or spinal cord. Scar tissue prevents nerve fibers from regenerating. This tissue is full of signal proteins that have an inhibitory effect: they instruct the fibers to stop growing. Wouldn't it be great if we could turn off this inhibitory effect until the nerve fibers have grown through the scar?”
Cancer
That is one use he intends to study, to investigate whether it is possible to switch the responsible receptor-ligand complex on and off with specific antibodies or chemicals. Another example is cancer. Signaling proteins play a role in the growth and spread of tumors by attracting or repelling cells. Jeroen says, “In that case, we would like to achieve the same by blocking the sensitivity of the tumor cells to those signaling proteins, possibly causing them to grow less rapidly and spread less far. These are two important applications; others include diseases such as ALS and defects in the immune system.”
Fundamental discovery
Contacts have already been established with companies to conduct further research, for example into nervous system damage. Discussions are also underway about a collaboration for the development and testing of specific antibodies. But, Jeroen wants to emphasize, this does not mean that new medicines will be available overnight. “It will take a while. This is a fundamental discovery at the very beginning of the medical science chain. It does show, however, how molecular biology can lay the foundations for possible new therapies for patients."
This research was funded by ALS Foundation Netherlands, Netherlands Organisation for Scientific Research (Vici grant ), Netherlands Organisation for Scientific Research and Ministry of Education, Culture and Science (Gravitation Program ‘Brainscapes), Stichting ParkinsonFonds and Utrecht University (research theme ‘Dynamics of Youth’).
